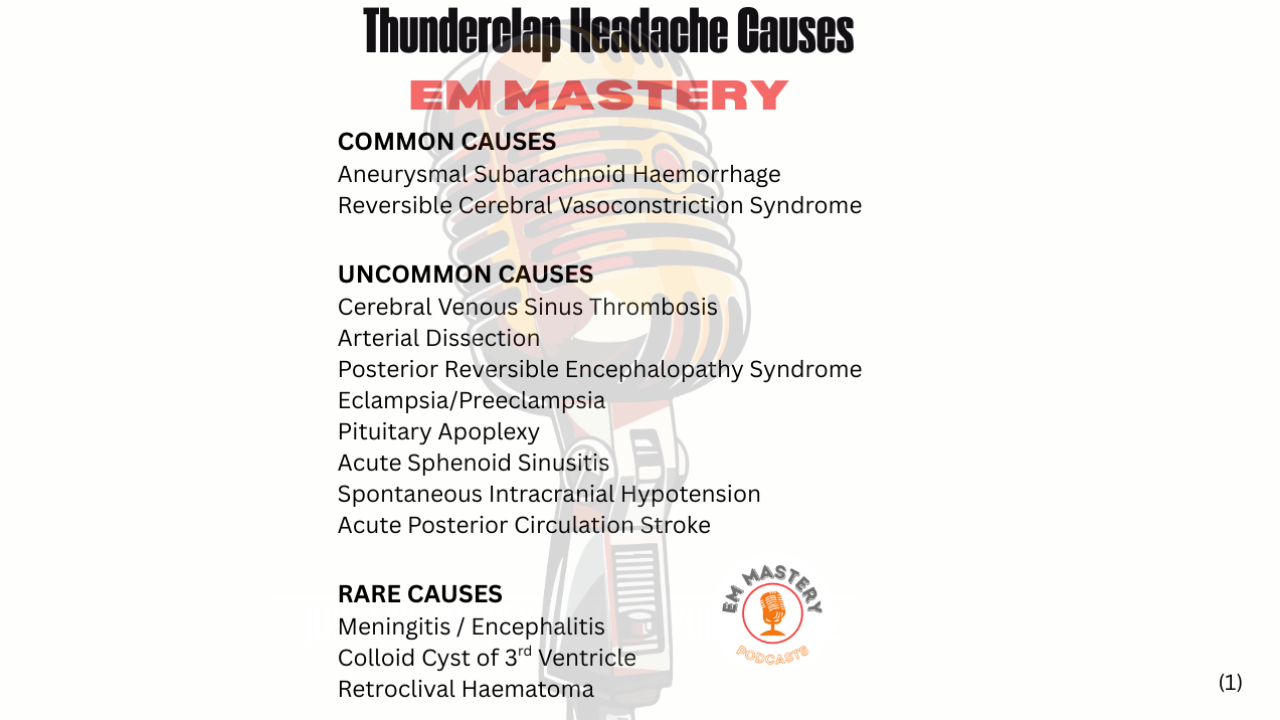
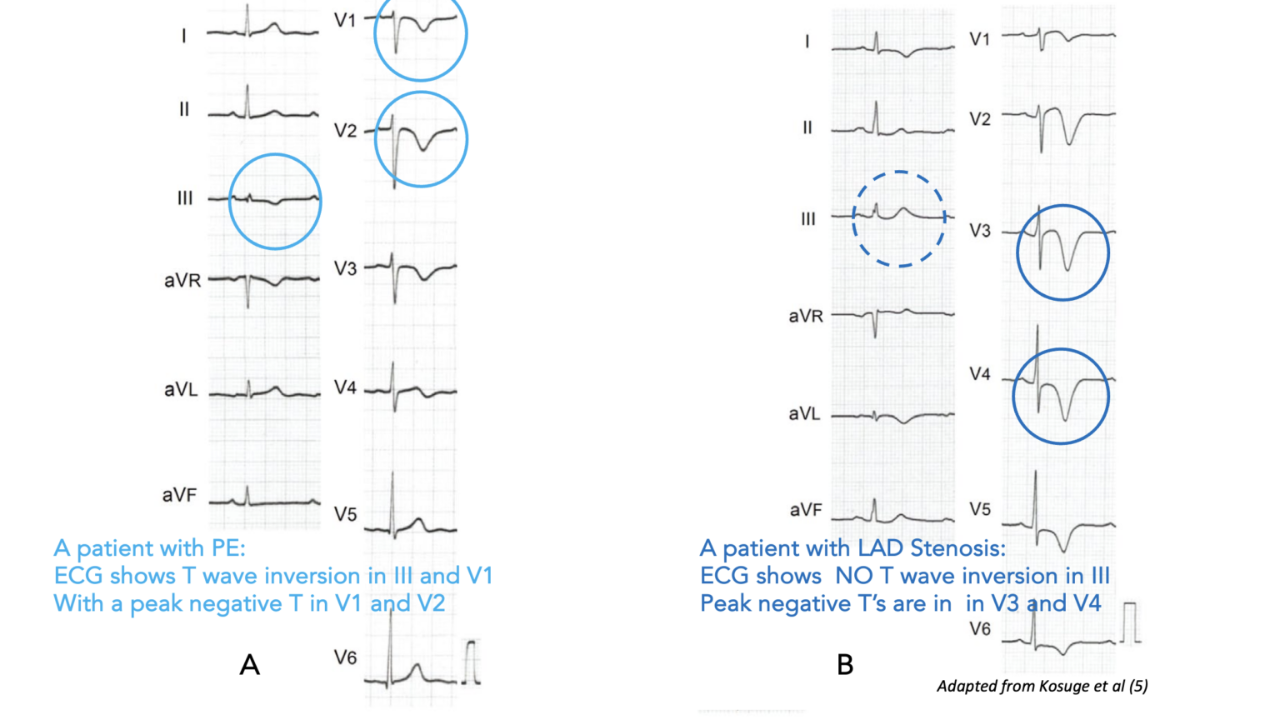
A Meta-analysis of NOAH-AFNET 6 and ARTESiA Trials
May 01, 2024Patients with subclinical atrial fibrillation, detected by implantable devices, have an increased stroke risk, although not as great as that of patient with clinical atrial fibrillation. I also include those patient with an incidental finding of atrial fibrillation in the emergency department ie., not only through interrogation of implantable devices.
What they did?
This study(1) performed a meta-analysis on two large randomised trials; the NOAH-AFNET 6 (2) and the ARTESiA (3) trials.
What they found
In terms of stroke outcomes:
- A reduction in a composite of all-cause stroke, peripheral arterial embolism, myocardial infarction, pulmonary embolism, or cardiovascular death with oral anticoagulation (RR, 0.85 [95% CI, 0.73–0.99]; I2=0%; Figure 2).
- A reduction in all-cause stroke (RR, 0.68 [95% CI, 0.51–0.90]; I2=0%), and a composite of all-cause stroke or systemic embolism with oral anticoagulation (RR, 0.65 [95% CI, 0.49–0.86]; I2 = 0%).
In terms of bleeding:
- An increase in major bleeding with oral anticoagulation (RR, 1.62 [95% CI, 1.05–2.5]; I2=61%
- There was no difference in fatal bleeding.
- The absolute risk increase for major bleeding was 7 more major bleeds per 1000 patient-years (95% CI, 1 more to 17 more).
Discussion
Anticoagulation with edoxaban or apixaban reduces stroke risk by one third and doubles major bleed risk. For me that is a significant bleeding risk unless the patient has stroke risk factors. However the rate of fatal bleeds was small.
If we find incidental atrial fibrillation in patients ie., they are asymptomatic, we should consider anticoagulation in in higher stroke risk patients. Does this translate to higher risk in those patients with low stroke risk criteria? Time will tell.
References
- McIntyre W F et al. Direct Oral Anticoagulants for Stroke Prevention in Patients With Device -Detected Atrial Fibrillation: A Study-Level Meta-Analysis of the NOAH-AFNET 6 and ARTESIA Trials. Circulation 2024;149:981-988
- Kirchhof P, et al. Probing oral anticoagu- lation in patients with atrial high rate episodes: rationale and design of the Non-Vitamin K Antagonist Oral Anticoagulants in Patients With Atrial High Rate Episodes (NOAH-AFNET 6) trial. Am Heart J. 2017;190:12–18.
- Lopes RD, et al. Rationale and design of the Apixaban for the Reduction of Thrombo-Embolism in Patients With Device-Detected Sub-Clinical Atrial Fibrillation (ARTESiA) trial. Am Heart J. 2017;189:137–145.
Join Our Free email updates
Get breaking news articles right in your inbox. Never miss a new article.
We hate SPAM. We will never sell your information, for any reason.